
Weak acid
Encyclopedia
A weak acid is an acid
that dissociates incompletely. It does not release all of its hydrogen
s in a solution, donating only a partial amount of its proton
s to the solution. These acids have higher pKa
than strong acids, which release all of their hydrogen atoms when dissolved in water.
Examples of weak acids include acetic acid
(CH3COOH) and oxalic acid
(H2C2O4).
in water solution
to only a moderate extent; that is, if the acid was represented by the general formula HA, then in aqueous solution a significant amount of undissociated HA still remains. Weak acids in water dissociate as:
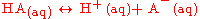
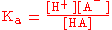
The strength of a weak acid is represented as either an equilibrium constant or as a percent dissociation. The equilibrium concentrations of reactants and products are related by the acid dissociation constant
expression, (Ka):
The greater the value of Ka, the more the formation of H+ is favored, and the lower the pH
of the solution. The Ka of weak acids varies between 1.8×10−16 and 55.5. Acids with a Ka less than 1.8×10−16 are weaker acids than water.
The other way to measure acid strength is to look at its percent dissociation, which is symbolized as α (alpha) and which can range from 0% < α < 100%. The percent dissociated is defined as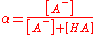
Unlike Ka, α is not constant and does depend on the [HA]. In general α will increase as [HA] decreases. Thus acids become stronger as they are diluted. If acids are polyprotic, then each proton will have a Ka. For example: H2CO3 + H2O → HCO3– + H3O+ has two Ka values because it has two acidic protons. The first Ka value is 4.46×10−7 (pKa1 = 6.351) and the second is 4.69×10−11 (pKa2 = 10.329).
 ), from the
), from the  of the acid (symbolized as HA), and by solving for concentration of H+ (symbolized by x and represented more accurately as H3O+). Below is a table that organizes the information. On the first line, the reaction is written. On the second line, the initial conditions are written below each compound. Note that a value of water is not given because its term (activity) in the
of the acid (symbolized as HA), and by solving for concentration of H+ (symbolized by x and represented more accurately as H3O+). Below is a table that organizes the information. On the first line, the reaction is written. On the second line, the initial conditions are written below each compound. Note that a value of water is not given because its term (activity) in the 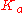 expression is technically equal to 1, but is often (conveniently) omitted. The third line shows how the value changes as the reaction goes to equilibrium. Then the last line gives the equilibrium concentrations and is simply the sum of each column.
expression is technically equal to 1, but is often (conveniently) omitted. The third line shows how the value changes as the reaction goes to equilibrium. Then the last line gives the equilibrium concentrations and is simply the sum of each column.
Applying the equilibrium line to the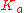 expression yields
expression yields
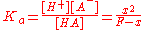
rearranging yields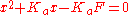 , which can be solved for x using the quadratic equation. The pH is then calculated as
, which can be solved for x using the quadratic equation. The pH is then calculated as  .
.

Solving for x yields

Then the pH = -log[H+]. The following equation then follows, but is only true if F >>> Ka
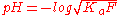
0.1M Solution
The full method gives the following quadratic:
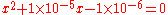
which gives x = 9.95×10−3 M and a pH = 3.00. The simplified method gives

So both methods yield the same result, but again F is more than 1000× greater than Ka. The next case does not have this condition and the results will differ.
5×10−4M Solution
The full method gives the following quadratic:
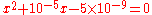
which gives x = 6.6×10−5 M and a pH = 4.18. The simplified method gives
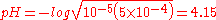
Here, the results differ by 0.03 pH units. As F becomes closer in value to the Ka, then the difference will increase even more. However, in practice, it is rare to work with such dilute acids and the pH is also dependent on ionic strength and temperature. So in reality, the simplified method works well.
s are generally assumed to be the most corrosive
, this is not always true. The carborane
superacid
(H(CHB11Cl11)), which is one million times stronger than sulfuric acid
, is entirely non-corrosive, whereas the weak acid hydrofluoric acid
(HF) is extremely corrosive and can dissolve, among other things, glass and all metals except iridium
.
Acid
An acid is a substance which reacts with a base. Commonly, acids can be identified as tasting sour, reacting with metals such as calcium, and bases like sodium carbonate. Aqueous acids have a pH of less than 7, where an acid of lower pH is typically stronger, and turn blue litmus paper red...
that dissociates incompletely. It does not release all of its hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
s in a solution, donating only a partial amount of its proton
Proton
The proton is a subatomic particle with the symbol or and a positive electric charge of 1 elementary charge. One or more protons are present in the nucleus of each atom, along with neutrons. The number of protons in each atom is its atomic number....
s to the solution. These acids have higher pKa
PKA
PKA, pKa, or other similar variations may stand for:* pKa, the symbol for the acid dissociation constant at logarithmic scale* Protein kinase A, a class of cAMP-dependent enzymes* Pi Kappa Alpha, the North-American social fraternity...
than strong acids, which release all of their hydrogen atoms when dissolved in water.
Examples of weak acids include acetic acid
Acetic acid
Acetic acid is an organic compound with the chemical formula CH3CO2H . It is a colourless liquid that when undiluted is also called glacial acetic acid. Acetic acid is the main component of vinegar , and has a distinctive sour taste and pungent smell...
(CH3COOH) and oxalic acid
Oxalic acid
Oxalic acid is an organic compound with the formula H2C2O4. This colourless solid is a dicarboxylic acid. In terms of acid strength, it is about 3,000 times stronger than acetic acid. Oxalic acid is a reducing agent and its conjugate base, known as oxalate , is a chelating agent for metal cations...
(H2C2O4).
Dissociation
Weak acids ionizeIonization
Ionization is the process of converting an atom or molecule into an ion by adding or removing charged particles such as electrons or other ions. This is often confused with dissociation. A substance may dissociate without necessarily producing ions. As an example, the molecules of table sugar...
in water solution
Solution
In chemistry, a solution is a homogeneous mixture composed of only one phase. In such a mixture, a solute is dissolved in another substance, known as a solvent. The solvent does the dissolving.- Types of solutions :...
to only a moderate extent; that is, if the acid was represented by the general formula HA, then in aqueous solution a significant amount of undissociated HA still remains. Weak acids in water dissociate as:
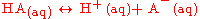
The strength of a weak acid is represented as either an equilibrium constant or as a percent dissociation. The equilibrium concentrations of reactants and products are related by the acid dissociation constant
Acid dissociation constant
An acid dissociation constant, Ka, is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction known as dissociation in the context of acid-base reactions...
expression, (Ka):

The greater the value of Ka, the more the formation of H+ is favored, and the lower the pH
PH
In chemistry, pH is a measure of the acidity or basicity of an aqueous solution. Pure water is said to be neutral, with a pH close to 7.0 at . Solutions with a pH less than 7 are said to be acidic and solutions with a pH greater than 7 are basic or alkaline...
of the solution. The Ka of weak acids varies between 1.8×10−16 and 55.5. Acids with a Ka less than 1.8×10−16 are weaker acids than water.
The other way to measure acid strength is to look at its percent dissociation, which is symbolized as α (alpha) and which can range from 0% < α < 100%. The percent dissociated is defined as
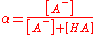
Unlike Ka, α is not constant and does depend on the [HA]. In general α will increase as [HA] decreases. Thus acids become stronger as they are diluted. If acids are polyprotic, then each proton will have a Ka. For example: H2CO3 + H2O → HCO3– + H3O+ has two Ka values because it has two acidic protons. The first Ka value is 4.46×10−7 (pKa1 = 6.351) and the second is 4.69×10−11 (pKa2 = 10.329).
Calculating the pH of a weak acid solution
The pH of a solution of a weak acid depends on the strength of the acid and the other components in the solution. In the simplest case, the weak acid is the only compound in water. In this case, the pH can be found from the concentration of the acid (symbolized as ), from the
), from the 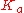 of the acid (symbolized as HA), and by solving for concentration of H+ (symbolized by x and represented more accurately as H3O+). Below is a table that organizes the information. On the first line, the reaction is written. On the second line, the initial conditions are written below each compound. Note that a value of water is not given because its term (activity) in the
of the acid (symbolized as HA), and by solving for concentration of H+ (symbolized by x and represented more accurately as H3O+). Below is a table that organizes the information. On the first line, the reaction is written. On the second line, the initial conditions are written below each compound. Note that a value of water is not given because its term (activity) in the 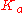 expression is technically equal to 1, but is often (conveniently) omitted. The third line shows how the value changes as the reaction goes to equilibrium. Then the last line gives the equilibrium concentrations and is simply the sum of each column.
expression is technically equal to 1, but is often (conveniently) omitted. The third line shows how the value changes as the reaction goes to equilibrium. Then the last line gives the equilibrium concentrations and is simply the sum of each column.
| HA(aq) | + | H2O(l) | → | A–(aq) | + | H3O+(aq) | |
| initial | F | — | 0 | 0 | |||
| change | -x | — | +x | +x | |||
| equilibrium | F - x | — | x | x |
Applying the equilibrium line to the
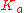 expression yields
expression yields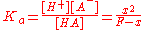
rearranging yields
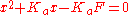 , which can be solved for x using the quadratic equation. The pH is then calculated as
, which can be solved for x using the quadratic equation. The pH is then calculated as  .
.Simplification
However, if F is more than 1000× greater than Ka, then (1) the acid will not deprotonate much, (2) the value of x will be small, and therefore (3) F - x ≈ F. This simplifies the Ka expression to...
Solving for x yields

Then the pH = -log[H+]. The following equation then follows, but is only true if F >>> Ka
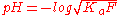
Comparison of the full and simplified methods
A certain weak acid has a Ka = 1×10−5 and the pH of two solutions needs to be found. One solution has a concentration of 0.10M and another has a concentration of 5×10−4M. The pH for both solutions will be calculated using both methods to yield 4 values, which will be compared.0.1M Solution
The full method gives the following quadratic:
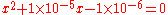
which gives x = 9.95×10−3 M and a pH = 3.00. The simplified method gives

So both methods yield the same result, but again F is more than 1000× greater than Ka. The next case does not have this condition and the results will differ.
5×10−4M Solution
The full method gives the following quadratic:
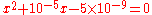
which gives x = 6.6×10−5 M and a pH = 4.18. The simplified method gives
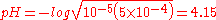
Here, the results differ by 0.03 pH units. As F becomes closer in value to the Ka, then the difference will increase even more. However, in practice, it is rare to work with such dilute acids and the pH is also dependent on ionic strength and temperature. So in reality, the simplified method works well.
Conjugate acid/base pair
It is often stated that "the conjugate of a weak acid is a strong base". This statement can be misleading. Most weak acids that textbooks discuss have weak (not strong) conjugate bases. Truly, only the very weakest of acids have strong conjugate bases. For example, if a weak acid has a Ka = 10−5, then its conjugate base would have a Kb = 10−9 (from the relationship Ka × Kb = 10−14), which certainly is not a strong base. A very weak acid with a Ka = 10−20 would indeed have a strong conjugate base.Corrosivity
While strong acidStrong acid
A strong acid is an acid that ionizes completely in an aqueous solution by losing one proton, according to the equationFor sulfuric acid which is diprotic, the "strong acid" designation refers only to dissociation of the first protonMore precisely, the acid must be stronger in aqueous solution than...
s are generally assumed to be the most corrosive
Corrosive
A corrosive substance is one that will destroy or irreversibly damage another surface or substance with which it comes into contact. The main hazards to people include damage to the eyes, the skin, and the tissue under the skin; inhalation or ingestion of a corrosive substance can damage the...
, this is not always true. The carborane
Carborane
A carborane is a cluster composed of boron and carbon atoms. Like many of the related boranes, these clusters are polyhedra and are similarly classified as closo-, nido-, arachno-, hypho-, etc...
superacid
Superacid
According to the classical definition superacid is an acid with an acidity greater than that of 100% pure sulfuric acid, which has a Hammett acidity function of −12. According to the modern definition, superacid is a medium, in which the chemical potential of the proton is higher than in pure...
(H(CHB11Cl11)), which is one million times stronger than sulfuric acid
Sulfuric acid
Sulfuric acid is a strong mineral acid with the molecular formula . Its historical name is oil of vitriol. Pure sulfuric acid is a highly corrosive, colorless, viscous liquid. The salts of sulfuric acid are called sulfates...
, is entirely non-corrosive, whereas the weak acid hydrofluoric acid
Hydrofluoric acid
Hydrofluoric acid is a solution of hydrogen fluoride in water. It is a valued source of fluorine and is the precursor to numerous pharmaceuticals such as fluoxetine and diverse materials such as PTFE ....
(HF) is extremely corrosive and can dissolve, among other things, glass and all metals except iridium
Iridium
Iridium is the chemical element with atomic number 77, and is represented by the symbol Ir. A very hard, brittle, silvery-white transition metal of the platinum family, iridium is the second-densest element and is the most corrosion-resistant metal, even at temperatures as high as 2000 °C...
.

