
Cyclodecapentaene
Encyclopedia
Cyclodecapentaene or [10]annulene is an annulene
with molecular formula C10H10. This organic compound
is a conjugated
10 pi electron cyclic
system and according to Huckel's rule
it should display aromaticity
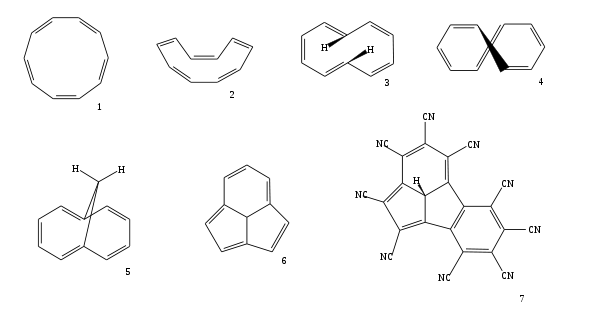
. It is not aromatic, however, because of a combination of steric strain and angular strain. The planar all-cis isomer (1) (like benzene is) would have bond angles of 144° which creates large amounts of bond strain relative to the ideal 120° for sp2 hybridization. A boat-like all-cis conformation (2) relieves much of the angular strain but is still unstable with respect to the next planar trans, cis, trans, cis, cis isomer (3). Yet this isomer suffers from steric repulsion between the two internal hydrogen atoms. The nonplanar trans, cis, cis, cis, cis isomer (4) is the most stable of all the possible isomers.
 The parent [10]annulene can be obtained by photolysis of cis-9,10-dihydronaphthalene as a mixture of isomers. Because of their lack in stability even at low temperatures the reaction products revert back to the original dihydronaphthalene.
The parent [10]annulene can be obtained by photolysis of cis-9,10-dihydronaphthalene as a mixture of isomers. Because of their lack in stability even at low temperatures the reaction products revert back to the original dihydronaphthalene.
Aromaticity can still be induced in [10]annulenes by fixation of the planar geometries and there exist two methods to bring it about. Replacing two hydrogen atoms by a methylene
bridge gives the planar bicyclic 1,6-methano[10]annulene (5). This compound is aromatic as judged from lack in bond length
alternation by x-ray crystallography
and a telltale diamagnetic ring current from NMR spectroscopy
.
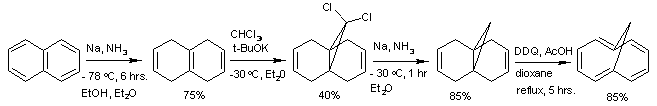 A classical organic synthesis
A classical organic synthesis
of this compound starts from a Birch reduction
of naphthalene
to tetrahydronaphthalene (or isodecalin), is followed by carbene
addition of dichlorocarbene
(prepared in-situ from chloroform
and the potassium salt of tert-butanol
) followed by a second organic reduction that removes the chloride substituent
s and is concluded by the organic oxidation by 2,3-dichloro-5,6-dicyano-1,4-benzoquinone
(DDQ).
Another way to restore aromaticity in [10]annulene by plane fixation is by incorporation of a methine
bridge to a tricyclic[10]annulene core structure (6) 2. When deprotonated
to form the anion this type of compound is even more stabilized. The central carbanion
makes the molecule even more planar and the number of resonance structures that can be drawn is extended to 7 included two resonance forms with a complete benzene ring. By computational chemistry
it is demonstrated that the tricyclic[10]annulene derivative with an annulated benzene ring and a full set of cyano substituent
s (7) is one of the most acidic compounds known with a computed pKa
in DMSO
of - 30.4 (compared to for instance -20 for magic acid
) 1.
Azulene
is also a 10 pi electron system in which aromaticity
is maintained but in yet another way.
Annulene
Annulenes are completely conjugated monocyclic hydrocarbons. They have the general formula CnHn or CnHn+1...
with molecular formula C10H10. This organic compound
Organic compound
An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. For historical reasons discussed below, a few types of carbon-containing compounds such as carbides, carbonates, simple oxides of carbon, and cyanides, as well as the...
is a conjugated
Conjugated system
In chemistry, a conjugated system is a system of connected p-orbitals with delocalized electrons in compounds with alternating single and multiple bonds, which in general may lower the overall energy of the molecule and increase stability. Lone pairs, radicals or carbenium ions may be part of the...
10 pi electron cyclic
Cyclic compound
In chemistry, a cyclic compound is a compound in which a series of atoms is connected to form a loop or ring.While the vast majority of cyclic compounds are organic, a few inorganic substances form cyclic compounds as well, including sulfur, silanes, phosphanes, phosphoric acid, and triboric acid. ...
system and according to Huckel's rule
Hückel's rule
In organic chemistry, Hückel's rule estimates whether a planar ring molecule will have aromatic properties. The quantum mechanical basis for its formulation was first worked out by physical chemist Erich Hückel in 1931...
it should display aromaticity
Aromaticity
In organic chemistry, Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization stronger than would be expected by the stabilization of conjugation alone. The earliest use of the term was in an article by August...
. It is not aromatic, however, because of a combination of steric strain and angular strain. The planar all-cis isomer (1) (like benzene is) would have bond angles of 144° which creates large amounts of bond strain relative to the ideal 120° for sp2 hybridization. A boat-like all-cis conformation (2) relieves much of the angular strain but is still unstable with respect to the next planar trans, cis, trans, cis, cis isomer (3). Yet this isomer suffers from steric repulsion between the two internal hydrogen atoms. The nonplanar trans, cis, cis, cis, cis isomer (4) is the most stable of all the possible isomers.

Aromaticity can still be induced in [10]annulenes by fixation of the planar geometries and there exist two methods to bring it about. Replacing two hydrogen atoms by a methylene
Methylene
Methylene is a chemical species in which a carbon atom is bonded to two hydrogen atoms. Three different possibilities present themselves:* the -CH2- substituent group: e.g., dichloromethane ....
bridge gives the planar bicyclic 1,6-methano[10]annulene (5). This compound is aromatic as judged from lack in bond length
Bond length
- Explanation :Bond length is related to bond order, when more electrons participate in bond formation the bond will get shorter. Bond length is also inversely related to bond strength and the bond dissociation energy, as a stronger bond will be shorter...
alternation by x-ray crystallography
X-ray crystallography
X-ray crystallography is a method of determining the arrangement of atoms within a crystal, in which a beam of X-rays strikes a crystal and causes the beam of light to spread into many specific directions. From the angles and intensities of these diffracted beams, a crystallographer can produce a...
and a telltale diamagnetic ring current from NMR spectroscopy
NMR spectroscopy
Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy, is a research technique that exploits the magnetic properties of certain atomic nuclei to determine physical and chemical properties of atoms or the molecules in which they are contained...
.

Organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the construction of organic compounds via organic reactions. Organic molecules can often contain a higher level of complexity compared to purely inorganic compounds, so the synthesis of organic compounds has...
of this compound starts from a Birch reduction
Birch reduction
The Birch Reduction is an organic reaction which is particularly useful in synthetic organic chemistry. The reaction was reported in 1944 by the Australian chemist Arthur Birch working in the Dyson Perrins Laboratory in the University of Oxford, building on earlier work by Wooster and Godfrey in...
of naphthalene
Naphthalene
Naphthalene is an organic compound with formula . It is a white crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 ppm by mass. As an aromatic hydrocarbon, naphthalene's structure consists of a fused pair of benzene rings...
to tetrahydronaphthalene (or isodecalin), is followed by carbene
Carbene
In chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is RR'C:, but the carbon can instead be double-bonded to one group. The term "carbene" may also merely refer to the compound H2C:, also called...
addition of dichlorocarbene
Dichlorocarbene
Dichlorocarbene is a carbene commonly encountered in organic chemistry. This reactive intermediate with chemical formula CCl2 is easily available by reaction of chloroform and a base such as potassium t-butoxide or sodium hydroxide dissolved in water...
(prepared in-situ from chloroform
Chloroform
Chloroform is an organic compound with formula CHCl3. It is one of the four chloromethanes. The colorless, sweet-smelling, dense liquid is a trihalomethane, and is considered somewhat hazardous...
and the potassium salt of tert-butanol
Tert-Butanol
tert-Butanol, or 2-methyl-2-propanol, is the simplest tertiary alcohol. It is one of the four isomers of butanol. tert-Butanol is a clear liquid with a camphor-like odor. It is very soluble in water and miscible with ethanol and diethyl ether...
) followed by a second organic reduction that removes the chloride substituent
Substituent
In organic chemistry and biochemistry, a substituent is an atom or group of atoms substituted in place of a hydrogen atom on the parent chain of a hydrocarbon...
s and is concluded by the organic oxidation by 2,3-dichloro-5,6-dicyano-1,4-benzoquinone
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone
2,3-Dichloro-5,6-dicyano-1,4-benzoquinone is the chemical reagent with formula C8Cl2N2O2. This oxidant is useful for the dehydrogenation of alcohols, phenols and steroid ketones in organic chemistry. DDQ decomposes in water, but is stable in aqueous mineral acid.-Preparation:Synthesis of DDQ...
(DDQ).
Another way to restore aromaticity in [10]annulene by plane fixation is by incorporation of a methine
Methine
In chemistry, methine is a trivalent functional group CH, derived formally from methane. The methine group consists of a carbon atom bound by two single bonds and one double bond, where one of the single bonds is to a hydrogen...
bridge to a tricyclic[10]annulene core structure (6) 2. When deprotonated
Deprotonation
Deprotonation is the removal of a proton from a molecule, forming the conjugate base.The relative ability of a molecule to give up a proton is measured by its pKa value. A low pKa value indicates that the compound is acidic and will easily give up its proton to a base...
to form the anion this type of compound is even more stabilized. The central carbanion
Carbanion
A carbanion is an anion in which carbon has an unshared pair of electrons and bears a negative charge usually with three substituents for a total of eight valence electrons. The carbanion exists in a trigonal pyramidal geometry. Formally a carbanion is the conjugate base of a carbon acid.where B...
makes the molecule even more planar and the number of resonance structures that can be drawn is extended to 7 included two resonance forms with a complete benzene ring. By computational chemistry
Computational chemistry
Computational chemistry is a branch of chemistry that uses principles of computer science to assist in solving chemical problems. It uses the results of theoretical chemistry, incorporated into efficient computer programs, to calculate the structures and properties of molecules and solids...
it is demonstrated that the tricyclic[10]annulene derivative with an annulated benzene ring and a full set of cyano substituent
Substituent
In organic chemistry and biochemistry, a substituent is an atom or group of atoms substituted in place of a hydrogen atom on the parent chain of a hydrocarbon...
s (7) is one of the most acidic compounds known with a computed pKa
PKA
PKA, pKa, or other similar variations may stand for:* pKa, the symbol for the acid dissociation constant at logarithmic scale* Protein kinase A, a class of cAMP-dependent enzymes* Pi Kappa Alpha, the North-American social fraternity...
in DMSO
Dimethyl sulfoxide
Dimethyl sulfoxide is an organosulfur compound with the formula 2SO. This colorless liquid is an important polar aprotic solvent that dissolves both polar and nonpolar compounds and is miscible in a wide range of organic solvents as well as water...
of - 30.4 (compared to for instance -20 for magic acid
Magic acid
Magic acid , is a superacid consisting of a mixture, most commonly in a 1:1 molar ratio, of fluorosulfonic acid and antimony pentafluoride...
) 1.
Azulene
Azulene
Azulene is an organic compound and an isomer of naphthalene. Whereas naphthalene is colourless, azulene is dark blue. Its name is derived from the Spanish word azul, meaning "blue"...
is also a 10 pi electron system in which aromaticity
Aromaticity
In organic chemistry, Aromaticity is a chemical property in which a conjugated ring of unsaturated bonds, lone pairs, or empty orbitals exhibit a stabilization stronger than would be expected by the stabilization of conjugation alone. The earliest use of the term was in an article by August...
is maintained but in yet another way.
External links
- 1,6-methano[10]annulene in Organic SynthesesOrganic SynthesesOrganic Syntheses is a scientific journal that since 1921 has provided the chemistry community with annual collections of detailed and checked procedures for the organic synthesis of organic compounds. The journal is peer reviewed...
, Coll. Vol. 6, p.731; Vol. 54, p.11 Online Article

