_oxide.gif)
Mercury(II) oxide
Encyclopedia
Mercury oxide, also called mercuric oxide or simply mercury oxide, has a formula of Hg
O
. It has a red or orange color. Mercury(II) oxide is a solid at room temperature and pressure. The mineral form montroydite is very rarely found.
discovered that oxygen was released by heating mercuric oxide, although he did not identify the gas as Oxygen
(rather, Priestley called it "dephlogisticated air", as that was the paradigm
that he was working under at the time.)
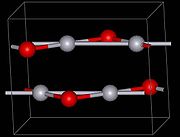
 The red form of HgO can be made by heating Hg on oxygen at roughly 350 °C, or by pyrolysis
The red form of HgO can be made by heating Hg on oxygen at roughly 350 °C, or by pyrolysis
of Hg(NO3)2
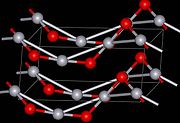
. The yellow form can be obtained by precipitation of aqueous Hg2+ with alkali. The difference in color is due to particle size, both forms have the same structure consisting of near linear O-Hg-O units linked in zigzag chains with an O-Hg-O angle of 108°.
(hexagonal
,
hP6, P3221); both are characterized by Hg-O chains. At pressures above 10 GPa both of those structures convert to a tetragonal form.
O
is sometimes used in the production of mercury as it decomposes quite easily. When it decomposes, oxygen gas is generated.
It is also used as a material for cathode
s for mercury batteries
.
 Mercury oxide is a toxic substance which can be absorbed into the body by inhalation of its aerosol, through the skin and by ingestion. The substance is irritating to the eyes, the skin and the respiratory tract and may have effects on the kidneys, resulting in kidney impairment. In the food chain important to humans, bioaccumulation takes place, specifically in aquatic organisms. The substance is banned as a pesticide in the EU.
Mercury oxide is a toxic substance which can be absorbed into the body by inhalation of its aerosol, through the skin and by ingestion. The substance is irritating to the eyes, the skin and the respiratory tract and may have effects on the kidneys, resulting in kidney impairment. In the food chain important to humans, bioaccumulation takes place, specifically in aquatic organisms. The substance is banned as a pesticide in the EU.
Evaporation at 20°C is negligible. HgO decomposes on exposure to light or on heating above 500°C. Heating produces highly toxic mercury fumes and oxygen, which increases the fire hazard. Mercury(II) oxide reacts violently with reducing agents, chlorine, hydrogen peroxide, magnesium (when heated), disulfur dichloride and hydrogen trisulfide. Shock-sensitive compounds are formed with metals and elements such as sulfur and phosphorus.
Mercury (element)
Mercury is a chemical element with the symbol Hg and atomic number 80. It is also known as quicksilver or hydrargyrum...
O
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
. It has a red or orange color. Mercury(II) oxide is a solid at room temperature and pressure. The mineral form montroydite is very rarely found.
History
In 1774, Joseph PriestleyJoseph Priestley
Joseph Priestley, FRS was an 18th-century English theologian, Dissenting clergyman, natural philosopher, chemist, educator, and political theorist who published over 150 works...
discovered that oxygen was released by heating mercuric oxide, although he did not identify the gas as Oxygen
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
(rather, Priestley called it "dephlogisticated air", as that was the paradigm
Paradigm
The word paradigm has been used in science to describe distinct concepts. It comes from Greek "παράδειγμα" , "pattern, example, sample" from the verb "παραδείκνυμι" , "exhibit, represent, expose" and that from "παρά" , "beside, beyond" + "δείκνυμι" , "to show, to point out".The original Greek...
that he was working under at the time.)
Synthesis


Pyrolysis
Pyrolysis is a thermochemical decomposition of organic material at elevated temperatures without the participation of oxygen. It involves the simultaneous change of chemical composition and physical phase, and is irreversible...
of Hg(NO3)2
Mercury(II) nitrate
Mercury nitrate is a toxic colorless or white soluble crystalline mercury salt of nitric acid. It was also used to treat fur to make felt in a process called 'carroting'. The phrase 'mad as a hatter' is associated with psychological illness brought on by excessive exposure to mercury nitrate...
. The yellow form can be obtained by precipitation of aqueous Hg2+ with alkali. The difference in color is due to particle size, both forms have the same structure consisting of near linear O-Hg-O units linked in zigzag chains with an O-Hg-O angle of 108°.
Structure
Under atmospheric pressure mercuric oxide has two crystalline forms: one is called montroydite (orthorhombic, 2/m 2/m 2/m, Pnma), and the second is analogous to the sulfide mineral cinnabarCinnabar
Cinnabar or cinnabarite , is the common ore of mercury.-Word origin:The name comes from κινναβαρι , a Greek word most likely applied by Theophrastus to several distinct substances...
(hexagonal
Hexagonal crystal system
In crystallography, the hexagonal crystal system is one of the 7 crystal systems, the hexagonal lattice system is one of the 7 lattice systems, and the hexagonal crystal family is one of the 6 crystal families...
,
hP6, P3221); both are characterized by Hg-O chains. At pressures above 10 GPa both of those structures convert to a tetragonal form.
Uses
HgMercury (element)
Mercury is a chemical element with the symbol Hg and atomic number 80. It is also known as quicksilver or hydrargyrum...
O
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
is sometimes used in the production of mercury as it decomposes quite easily. When it decomposes, oxygen gas is generated.
It is also used as a material for cathode
Cathode
A cathode is an electrode through which electric current flows out of a polarized electrical device. Mnemonic: CCD .Cathode polarity is not always negative...
s for mercury batteries
Mercury battery
A mercury battery is a non-rechargeable electrochemical battery, a primary cell. Due to the content of mercury, and the resulting environmental concerns, the sale of mercury batteries is banned in many countries. Both ANSI and IEC have withdrawn standards for mercury batteries...
.
Health issues

Evaporation at 20°C is negligible. HgO decomposes on exposure to light or on heating above 500°C. Heating produces highly toxic mercury fumes and oxygen, which increases the fire hazard. Mercury(II) oxide reacts violently with reducing agents, chlorine, hydrogen peroxide, magnesium (when heated), disulfur dichloride and hydrogen trisulfide. Shock-sensitive compounds are formed with metals and elements such as sulfur and phosphorus.

