Epimer
Encyclopedia
In chemistry
, epimers are diastereomers that differ in configuration of only one stereogenic center. Diastereomers are a class of stereoisomers that are non-superposable, non-mirror images of one another.
In chemical nomenclature, one of the epimeric pairs is given the prefix epi- for example in quinine
and epi-quinine. When the pairs are enantiomers, the prefix becomes ent-.
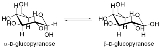
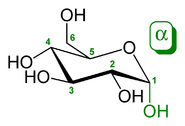
s α-glucose and β-glucose are epimers. In α-glucose, the -OH group on the first (anomeric) carbon is in the direction opposite the methylene
group on carbon C-6 (in the axial position). In β-glucose, the -OH group is oriented in the same direction as the methylene group (in the equatorial position). These two molecules are both epimers and anomers.
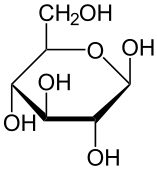
β-D-glucopyranose and β-D-mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position. The hydroxyl group in β-D-glucopyranose is equatorial (in the "plane" of the ring) while in β-D-mannopyranose the C-2 hydroxyl group is axial (up from the "plane" of the ring). These two molecules are epimers but not anomer
s.
Doxorubicin
and epirubicin
are two closely related drugs and epimers.
Other closely related compounds are epi-inositol
and inositol
and lipoxin
and epilipoxin.
.
Chemistry
Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry is concerned with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds....
, epimers are diastereomers that differ in configuration of only one stereogenic center. Diastereomers are a class of stereoisomers that are non-superposable, non-mirror images of one another.
In chemical nomenclature, one of the epimeric pairs is given the prefix epi- for example in quinine
Quinine
Quinine is a natural white crystalline alkaloid having antipyretic , antimalarial, analgesic , anti-inflammatory properties and a bitter taste. It is a stereoisomer of quinidine which, unlike quinine, is an anti-arrhythmic...
and epi-quinine. When the pairs are enantiomers, the prefix becomes ent-.
Examples
The sugarSugar
Sugar is a class of edible crystalline carbohydrates, mainly sucrose, lactose, and fructose, characterized by a sweet flavor.Sucrose in its refined form primarily comes from sugar cane and sugar beet...
s α-glucose and β-glucose are epimers. In α-glucose, the -OH group on the first (anomeric) carbon is in the direction opposite the methylene
Methylene
Methylene is a chemical species in which a carbon atom is bonded to two hydrogen atoms. Three different possibilities present themselves:* the -CH2- substituent group: e.g., dichloromethane ....
group on carbon C-6 (in the axial position). In β-glucose, the -OH group is oriented in the same direction as the methylene group (in the equatorial position). These two molecules are both epimers and anomers.
 |
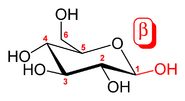 |
β-D-glucopyranose and β-D-mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position. The hydroxyl group in β-D-glucopyranose is equatorial (in the "plane" of the ring) while in β-D-mannopyranose the C-2 hydroxyl group is axial (up from the "plane" of the ring). These two molecules are epimers but not anomer
Anomer
In carbohydrate chemistry, an anomer is a special type of epimer. It is one of two stereoisomers of a cyclic saccharide that differs only in its configuration at the hemiacetal or hemiketal carbon, also called the anomeric carbon. Anomerization is the process of conversion of one anomer to the other...
s.
 |
|
| |
Doxorubicin
Doxorubicin
Doxorubicin INN is a drug used in cancer chemotherapy. It is an anthracycline antibiotic, closely related to the natural product daunomycin, and like all anthracyclines, it works by intercalating DNA....
and epirubicin
Epirubicin
Epirubicin is an anthracycline drug used for chemotherapy. It is marketed by Pfizer under the trade name Ellence in the US and Pharmorubicin or Epirubicin Ebewe elsewhere....
are two closely related drugs and epimers.
Other closely related compounds are epi-inositol
Epi-inositol
epi-Inositol is one of the stereoisomers of inositol.-See also:*allo-Inositol*cis-Inositol*D-chiro-Inositol*L-chiro-Inositol*muco-Inositol*neo-Inositol*scyllo-Inositol...
and inositol
Inositol
Inositol or cyclohexane-1,2,3,4,5,6-hexol is a chemical compound with formula 6126 or 6, a sixfold alcohol of cyclohexane. It exists in nine possible stereoisomers, of which the most prominent form, widely occurring in nature, is cis-1,2,3,5-trans-4,6-cyclohexanehexol, or myo-inositol...
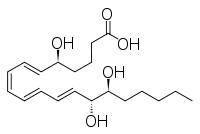
and lipoxin
Lipoxin
Lipoxins are a series of anti-inflammatory mediators. Lipoxins are short lived endogenously produced nonclassic eicosanoids whose appearance in inflammation signals the resolution of inflammation....
and epilipoxin.
 |
|||
Epimerisation
Epimerisation is a chemical process where an epimer is transformed into its chiral counterpart. It can happen in condensed tannins depolymerisation reactions. Epimersation can be spontaneous (generally a slow process), or catalyzed by enzymes, e.g. the epimerization between the sugars N-acetylglucosamine and N-acetylmannosamine, which is catalyzed by RENBPRENBP
N-acylglucosamine 2-epimerase is an enzyme that in humans is encoded by the RENBP gene.-Further reading:...
.

